Corcept (CORT) Posts Preliminary '20 Results, Gives 2021 View
Shares of Corcept Therapeutics Incorporated CORT were down 6.7% in after-hours trading on Monday after the company reported preliminary results for fourth-quarter and full-year 2020. The company also issued revenue guidance for 2021.
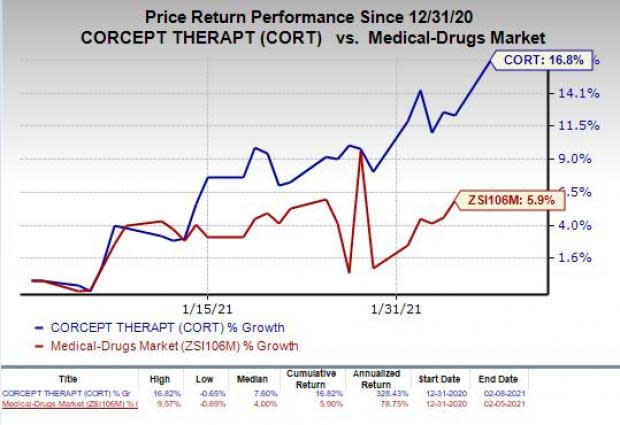
However, the stock has rallied 16.8% so far this year compared with the industry’s increase of 5.9%.
2020 Preliminary Results
Corcept reported preliminary fourth-quarter revenues of $85.7 million which reflect a decrease of 2.5% year over year. However, for full-year 2020, the company reported preliminary revenues of $353.9 million which reflect an increase of 15.5% year over year. As of Dec 31, 2020, preliminary cash and investments were $476.9 million.
Corcept’s only marketed drug, Korlym, is approved for the once-daily oral treatment of hyperglycemia — secondary to hypercortisolism — in adult patients with endogenous Cushing’s syndrome who are suffering from type II diabetes or glucose intolerance and who have already failed surgery or are unsuitable for the same.
Meanwhile, Corcept’s lead candidate, relacorilant, is being evaluated in the phase III GRACE study to treat Cushing’s syndrome. Dosing is currently underway in the above-mentioned study at sites across the United States, Canada, Europe and Israel. Enrollment is also underway in the phase III study of GRADIENT on relacorilant in patients whose Cushing’s syndrome is caused by adrenal adenoma at sites across the United States, Europe and Israel.
Owing to the adverse effects of the ongoing COVID-19 pandemic, timing of the new drug application filing for relacorilant is now expected to be in the second quarter of 2023. It was earlier expected that the filing would be done in the second quarter of 2022.
Corcept has completed enrollment in a phase II study on relacorilant in combination with Bristol Myers’ BMY Abraxane for the treatment of metastatic ovarian cancer. The combo of relacorilant plus Abraxane is also being evaluated for addressing patients with metastatic pancreatic cancer. Preliminary results from both studies are expected in first-half 2021.
Corcept is also evaluating relacorilant plus Merck’s MRK PD-1 checkpoint inhibitor Keytruda to address patients with metastatic or unresectable adrenal cancer.
Apart from relacorilant, Corcept’s other pipeline candidates include exicorilant and miricorilant.
The company is conducting an open-label, dose-finding study on exicorilant in combination with Pfizer’s PFE Xtandi for treating patients with metastatic castration-resistant prostate cancer. Selection of optimum dose in the study is expected by third quarter 2021.
Corcept is enrolling patients in a phase II GRATITUDE study on miricorilant for addressing the reversal of antipsychotic-induced weight gain (“APIWG”). Enrollment is also underway in the phase II GRATITUDE II study of miricorilant to reverse the long-standing APIWG. Meanwhile, in the fourth quarter of 2020, the company initiated a phase II study, evaluating miricorilant as a potential treatment for NASH, a serious liver disorder.
2021 Guidance
The company expects revenues of $375 – 405 million in 2021. The Zacks Consensus Estimate for the same stands at $408.1 million.
Zacks Rank
Corcept currently carries a Zacks Rank #4 (Sell). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
More Stock News: This Is Bigger than the iPhone!
It could become the mother of all technological revolutions. Apple sold a mere 1 billion iPhones in 10 years but a new breakthrough is expected to generate more than 77 billion devices by 2025, creating a $1.3 trillion market.
Zacks has just released a Special Report that spotlights this fast-emerging phenomenon and 4 tickers for taking advantage of it. If you don't buy now, you may kick yourself in 2022.
Click here for the 4 trades >>
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Merck & Co., Inc. (MRK) : Free Stock Analysis Report
To read this article on Zacks.com click here.
