LEXX: DehydraTECH CBD HYPER-H21-4 Results
NASDAQ:LEXX
READ THE FULL LEXX RESEARCH REPORT
HYPER-H21-4 Results
In an October 27th press release, Lexaria Bioscience Corporation (NASDAQ:LEXX) reported impressive results from its human clinical hypertension study designated HYPER-H21-4. This is the fourth and most comprehensive of Lexaria’s hypertension studies and was structured as a randomized, double-blinded, placebo-controlled, cross-over study that dosed 66 male and female volunteers between the ages of 40-70. The study compared DehydraTECH cannabidiol (CBD) against a placebo. Enrollment criteria sought patients with elevated blood pressure, stage 1 hypertension or stage 2 hypertension.1 Data were cataloged over the five weeks of dosing after recording baseline metrics. The first blood pressure measurement recorded to determine clinical endpoints was conducted over a 24-hour period at the end of the first 2.5 weeks and the second over a 24-hour period at the end of the second 2.5 weeks.
Results from the study showed an improvement in all time periods and for both systolic and diastolic pressures for subjects administered DehydraTECH CBD. Measurements of systolic and diastolic pressure for the first and second part of the dosing period compared to baseline ranged from 2.3 to 5.6 mm Hg.

Safety was a strong point for DehydraTECH CBD with eight mild to moderate adverse events recorded in the active arm compared to six mild to moderate events in the placebo arm. No serious adverse events occurred. Liver enzymes were within normal ranges and there were no adverse hepatic changes reported.
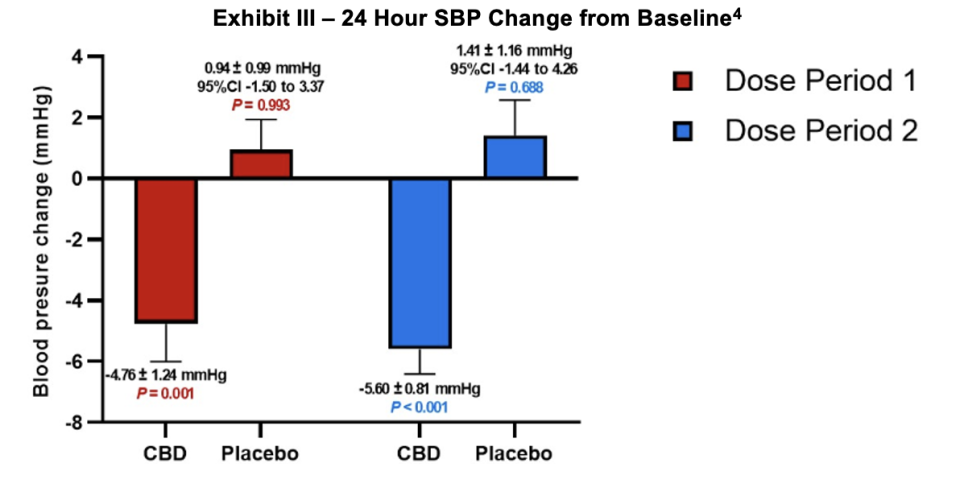
Additional graphical data was provided regarding the mean arterial pressure (MAP) compared to baseline. The data shows a statistically significant improvement compared with baseline and a 3.22 mm Hg lower reading for the DehydraTECH arm relative to baseline during the first half of the dosing period. In the second half of the dosing period, the difference was even greater where DehydraTECH also generated a statistically significant improvement relative to baseline of 4.14 mm Hg. The data is graphically represented in the following exhibit.

Systolic blood pressure (SBP) was reduced relative to baseline and placebo. Reductions were statistically significant across the board for each data segment and improvements relative to baseline were 4.76 and 5.60 mm Hg for the first and second halves of the dosing period respectively. Higher systolic blood pressure is the measure which has been most associated with a greater risk of stroke and heart disease.
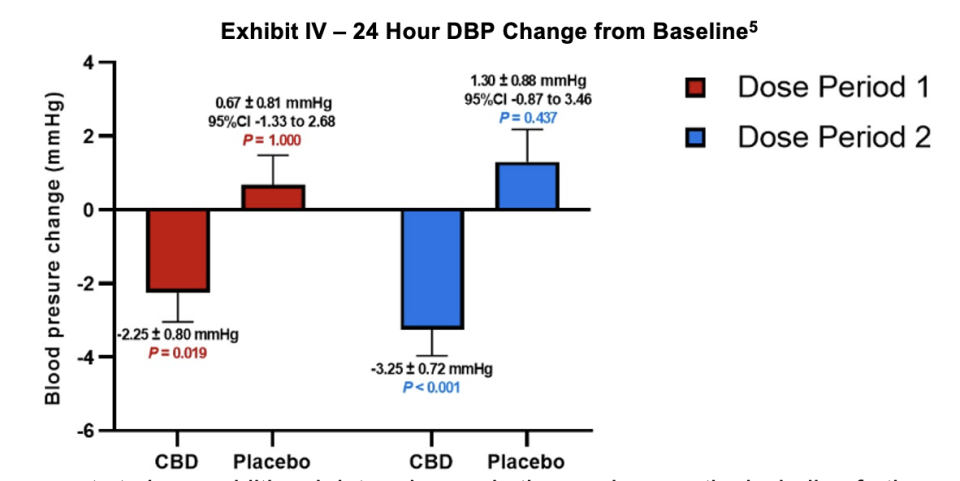
Lexaria also provided a graphic illustrating the comparative performance of patients’ diastolic blood pressure (DBP). This metric was reduced for the DehydraTECH CBD arm by 2.25 and 3.25 mm Hg relative to baseline for the first 2.5 and second 2.5 week periods.
Lexaria expects to have additional data releases in the coming months including further examinations of performance of subpopulations within the evaluated group and qualitative assessments based on questionnaire responses. A peer reviewed article has been developed based on available data and will be in preparation to be published in a journal over the coming months. Next steps for the hypertension program are to conduct a Phase Ib study which is targeted to begin in 2023.
The data generated to date has demonstrated early safety and efficacy for DehydraTECH CBD and we anticipate that the upcoming Phase Ib study will continue the positive results as it has a similar design to HYPER-H21-4. Longer term goals for the program are to find a partner who will advance the work to a registrational study and send it on the FDA for approval. While this is several years away, the data that has been released so far is supportive of further development.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
1. Elevated blood pressure was defined as systolic and diastolic pressures of 120/80 to 139/80 mm Hg, stage 1 from 140/90 to 159/99 mm Hg and stage 2 of 160/100 to 179/109 mm Hg.
2. Source: Lexaria Biosciences Press Release October 27, 2022.
3. Source: Lexaria Biosciences Press Release October 27, 2022.
4. Source: Lexaria Biosciences Press Release October 27, 2022.
5. Source: Lexaria Biosciences Press Release October 27, 2022.
