Rhythm (RYTM) Surges on Upbeat Data on Lead Product Candidate
Shares of Rhythm Phramaceuticals RYTM rallied 42.2% on Jul 12 after the company announced positive results from a phase II study on its lead product candidate, setmelanotide. The candidate will be used for treating severe obesity and hyperphagia in people with hypothalamic obesity.
Hypothalamic obesity is an extreme form of obesity that occurs due to damage to the brain’s hypothalamus. The symptoms of hypothalamic obesity may vary but mostly include uncontrollable hunger and rapid, excessive weight gain
The phase II study is a multi-center, open-label, proof-of-concept study evaluating the efficacy of setmelanotide for 16 weeks, in 18 patients between 6 and 28 years of age, with hypothalamic obesity. As of May 6, 2022, 11 patients were evaluable for assessment, including nine patients who completed the 16-week treatment and two patients who, due to treatment-related adverse events, discontinued early.
The primary endpoint of the study was to achieve more than a 5% reduction in BMI from baseline in evaluable patients, at 16 weeks, on setmelaotide therapy.
Setmelanotide also achieved a meaningful reduction in hunger scores.
Based on positive results from this mid-stage study, Rhythm intends to proceed to phase III clinical development, following consultations with regulatory agencies.
Shares of Rhythm have declined 33.1% so far this year compared with the industry’s fall of 19.7%.
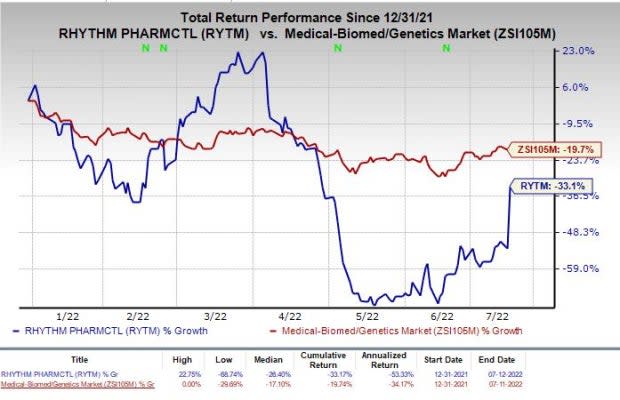
Image Source: Zacks Investment Research
Rhythm’s lead product candidate, setmelanotide, for chronic weight management, marketed under the brand name, Imcivree, received its FDA approval in November 2020.
The drug was approved for chronic weight management in adult and pediatric patients 6 years of age and older with obesity due to proopiomelanocortin (POMC), proprotein convertase subtilisin/kexin type 1 (PCSK1) or leptin receptor (LEPR) deficiency confirmed by genetic testing.
Recently, FDA approved Rhythm’s supplemental New Drug Application (sNDA), seeking the label expansion of Imcivree to include patients with Bardet-Biedl syndrome (BBS), a rare genetic obesity disease.
Following the recent approval, the drug has become the first and only FDA-approved treatment for BBS patients.
However, the FDA issued a complete response letter (CRL) for another sNDA seeking label expansion of Imcivree for the treatment of patients with Alström syndrome, another genetic disease that can cause obesity.
Rhythm Pharmaceuticals, Inc. Price
Rhythm Pharmaceuticals, Inc. price | Rhythm Pharmaceuticals, Inc. Quote
Zacks Rank & Stocks to Consider
Rhythm Pharmaceuticals currently carries a Zacks Rank #3 (Hold). Some similar ranked stocks in the overall health sector are Aquestive Therapeutics AQST, Merck & Co. MRK., and Seagen SGEN, each carrying a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
Aquestive Therapeutics’ loss per share estimates for 2022 have narrowed from $1.50 to $1.34 cents in the past 30 days. The same for 2023 has narrowed from 95 cents to 74 cents in the same time frame.
Earnings of Aquestuve missed estimates in one of the trailing four quarters and beat the same on the remaining three occasions, the average surprise being 13.78%.
Merck’s earnings per share estimates for 2022 have improved from $7.28 to $7.31 in the past 30 days. The same for 2023 has remained steady at $7.16,in the same time frame. Shares of MRK have returned 24.2% in the year-to-date period.
Earnings of Merck missed estimates in one of the trailing four quarters and beat the same on the remaining three occasions, the average surprise being 13.42%.
Seagen’s loss per share estimates for 2022 have narrowed from $3.50 to $3.49 in the past 30 days. The same for 2023 has widened from $1.41 to $1.43 cents in the same time frame. %. SGEN have returned 15.8% in the year-to-date period.
Earnings of Seagen missed estimates in two of the trailing four quarters and beat the same on the remaining two occasions, the average negative surprise being 40.08%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Merck & Co., Inc. (MRK) : Free Stock Analysis Report
Seagen Inc. (SGEN) : Free Stock Analysis Report
Rhythm Pharmaceuticals, Inc. (RYTM) : Free Stock Analysis Report
Aquestive Therapeutics, Inc. (AQST) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research
