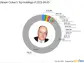
Iterum Therapeutics plc (ITRM)
- Previous Close
1.5800 - Open
1.6000 - Bid 1.4900 x 100
- Ask 1.6000 x 100
- Day's Range
1.5100 - 1.6000 - 52 Week Range
0.6220 - 2.5000 - Volume
35,939 - Avg. Volume
624,164 - Market Cap (intraday)
25.79M - Beta (5Y Monthly) 2.36
- PE Ratio (TTM)
-- - EPS (TTM)
-2.9200 - Earnings Date May 10, 2024 - May 14, 2024
- Forward Dividend & Yield --
- Ex-Dividend Date --
- 1y Target Est
6.50
Iterum Therapeutics plc, a clinical-stage pharmaceutical company, engages in developing and commercializing anti-infectives in Ireland, Bermuda, and the United States. It is developing sulopenem, a novel anti-infective compound with oral and IV formulations that is in Phase III clinical trials for the treatment of uncomplicated urinary tract infections, complicated urinary tract infections, and complicated intra-abdominal infections. The company was incorporated in 2015 and is headquartered in Dublin, Ireland.
www.iterumtx.comRecent News: ITRM
Performance Overview: ITRM
Trailing total returns as of 4/24/2024, which may include dividends or other distributions. Benchmark is .
YTD Return
1-Year Return
3-Year Return
5-Year Return
Compare To: ITRM
Select to analyze similar companies using key performance metrics; select up to 4 stocks.
Statistics: ITRM
Valuation Measures
Market Cap
25.79M
Enterprise Value
20.82M
Trailing P/E
--
Forward P/E
--
PEG Ratio (5yr expected)
--
Price/Sales (ttm)
--
Price/Book (mrq)
6.03
Enterprise Value/Revenue
--
Enterprise Value/EBITDA
-0.60
Financial Highlights
Profitability and Income Statement
Profit Margin
0.00%
Return on Assets (ttm)
-63.74%
Return on Equity (ttm)
-355.35%
Revenue (ttm)
--
Net Income Avi to Common (ttm)
-38.37M
Diluted EPS (ttm)
-2.9200
Balance Sheet and Cash Flow
Total Cash (mrq)
23.93M
Total Debt/Equity (mrq)
--
Levered Free Cash Flow (ttm)
-22.38M