Amicus (FOLD) Gets FDA Approval for Pompe Disease Drug
Amicus Therapeutics FOLD announced that the FDA approved Pombiliti (cipaglucosidase alfa) + Opfolda (miglustat) 65mg capsules, a two-component therapy for treating late-onset Pompe disease (LOPD) in adults, who weigh over 40 kgand are not improving on their current enzyme replacement therapy.
Cipaglucosidase alfa is a long-term enzyme replacement therapy and miglustat is an enzyme stabilizer component.
The approval presents a significant commercial opportunity for FOLD. Management will move forward with the launch of Pombiliti + Opfolda in the United States.
Shares of Amicus have nosedived 2.8% year to date compared with the industry’s 16.0% decline.
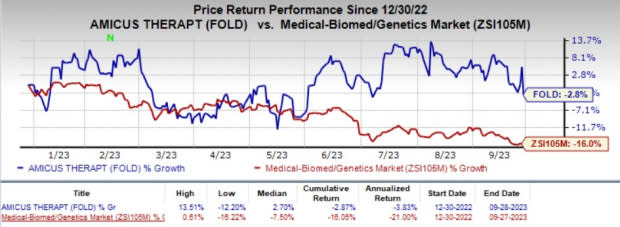
Image Source: Zacks Investment Research
The FDA approval was based on positive data from the phase III PROPEL study, the only randomized, controlled trial in LOPD. The study included patients in the high unmet need ERT-experienced population, in addition to ERT-naïve patients.
Pompe disease is a lysosomal disorder, resulting from the deficiency of the enzyme acid alpha-glucosidase. The disease is characterized by severe muscle weakness that worsens over time.
Earlier in June, Pombiliti + Opfolda also received approval in the Europe Union for treating LOPD in adults. Last month, Pombiliti + Opfolda was approved in the United Kingdom for the same indication. Approval of the drug in additional geographies could boost FOLD’s top line in the coming years.
In February 2019, the FDA granted Pombiliti + Opfolda the Breakthrough Therapy designation — the first ever granted to an investigational product for Pompe disease. The designation provides FOLD with additional market exclusivity and expedites development and review of drugs.
The company currently has one marketed drug in its portfolio, Galafold (migalastat), the first and only approved oral precision medicine for patients with Fabry disease with amenable genetic variants. In the first half of 2023, Galafold generated revenues of $180.8 million, up 13% year over year.
Amicus Therapeutics, Inc. Price and Consensus
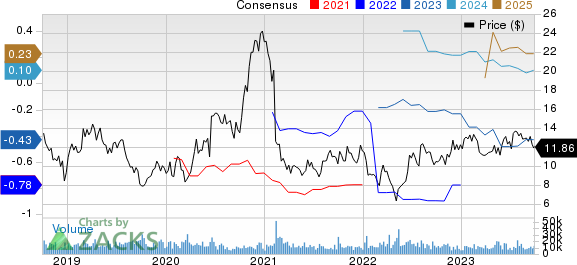
Amicus Therapeutics, Inc. price-consensus-chart | Amicus Therapeutics, Inc. Quote
Zacks Rank & Stocks to Consider
Amicus currently carries a Zacks Rank #3 (Buy).
Some better-ranked stocks in the same industry are Anika Therapeutics ANIK, Annovis Bio ANVS and Corcept Therapeutics CORT, each carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
In the past 90 days, the Zacks Consensus Estimate forAnika Therapeutics has narrowed from a loss of $1.41 per share to a loss of $1.24 for 2023. The bottom-line estimate has widened from a loss of 79 cents to a loss of 82 cents for 2024 during the same time frame. Shares of the company have lost 36.2% year to date.
ANIK’s earnings beat estimates in one of the trailing four quarters and missed the mark in the remaining three, delivering an average negative surprise of 32.12%.
In the past 90 days, the Zacks Consensus Estimate for Annovis Bio has narrowed from a loss of $4.89 per share to a loss of $4.38 for 2023. The bottom-line estimate has narrowed from a loss of $3.18 to a loss of $2.77 for 2024 during the same time frame. Shares of the company have lost 30.2% year to date.
ANVS’ earnings beat estimates in three of the trailing four quarters and missed the mark in one, delivering an average surprise of 13.40%.
In the past 90 days, the Zacks Consensus Estimate for Corcept’s earnings has gone up from 62 cents per share to 78 cents for 2023. The bottom-line estimate has also improved from 61 cents to 83 cents for 2024 during the same time frame. Shares of the company have rallied 62.3% year to date.
CORT’s earnings beat estimates in two of the trailing four quarters and missed the mark in the other two, delivering an average surprise of 6.99%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Amicus Therapeutics, Inc. (FOLD) : Free Stock Analysis Report
Corcept Therapeutics Incorporated (CORT) : Free Stock Analysis Report
Anika Therapeutics Inc. (ANIK) : Free Stock Analysis Report
Annovis Bio, Inc. (ANVS) : Free Stock Analysis Report
