Aptamer Sciences Inc. Files IND Application for Innovative Liver Cancer Treatment AST-201: A Promising Approach to Address Under Medical Needs
SEONGNAM, South Korea, March 25, 2024 /PRNewswire/ -- Aptamer Sciences Inc. (KOSDAQ, 291650) announced the filing of an Investigational New Drug (IND) application for the phase 1 clinical trial of AST-201 with the Korean Ministry of Food and Drug Safety on March 12th, 2024.
AST-201, the company's pioneering candidate in its pipeline, is an ApDCTM (Aptamer-Drug Conjugate) designed to target GPC3 protein, which exhibits high expression levels in specific cancer cells, notably liver cancer. It comprises gemcitabine as the payload and an aptamer that selectively binds to GPC3 protein. Gemcitabine, a proven systemic anticancer agent, is widely utilized in treating various cancers, including ovarian, breast, non-small cell lung, and pancreatic cancer. The choice of gemcitabine as the payload was deliberate, owing to its moderate toxicity profile, particularly suitable for liver targeting, given the organ's susceptibility to drug toxicity. Additionally, gemcitabine's nature as a DNA analogue facilitates its synthesis within the aptamer DNA sequence. The company's strategic focus on liver cancer addresses significant unmet medical needs, as evidenced by AST-201's superior efficacy over sorafenib in animal models and its synergistic effect when co-administered with anti-PD-1 therapy.
Liver cancer poses a significant health challenge, particularly in East Asia, with a dismal survival rate. While combination therapies such as atezolizumab and bevacizumab have emerged as standard treatments for advanced liver cancer, recent findings published in Nature Medicine suggest their limited efficacy in certain liver cancer populations with high GPC3 expression levels, highlighting substantial unmet medical needs. In this context, AST-201 offers a promising therapeutic approach by specifically targeting GPC3.
The planned phase 1 trial seeks to evaluate the safety, pharmacokinetic profile, and preliminary efficacy of AST-201 in patients with GPC3-positive advanced solid tumors. Notably, Dr. Hong Jae Chon from CHA Bundang Medical Center, a prominent medical oncologist in South Korea, will serve as the coordinating investigator. The trial will be conducted across four hospitals, including CHA Bundang Medical Center, Samsung Medical Center, Severance Hospital, and the National Cancer Center in South Korea.
Dr. Chon remarked, "GPC3 has garnered considerable attention in liver cancer therapeutic development among biopharmaceutical companies," expressing optimism that "AST-201 may emerge as a novel treatment option for liver cancer patients."
Aptamer Sciences Inc. aims to showcase the clinical benefits of AST-201 and the technological value of its ApDCTM platform through this trial. Moreover, the company anticipates that this milestone will propel its research and development efforts, enhancing the competitiveness of its subsequent hemato-oncology pipeline products, including AST-202 targeting CD25, ApRC (Aptamer Radioligand Conjugate), ApIS (Aptamer Immune Stimulator Conjugate), and so on.
Dr. Dong-il Han, CEO of Aptamer Sciences Inc., emphasized, "Since the second aptamer drug Izervay approved by FDA last year, aptamer technology has garnered significant attention in the pharmaceutical market," highlighting, "The AST-201 clinical trial will showcase the technological capabilities of the ApDCTM platform and expedite ongoing technology transfer discussion with multiple partners. This milestone underscores Aptamer Sciences Inc.'s commitment to advancing innovative therapies and addressing critical unmet needs in oncology."
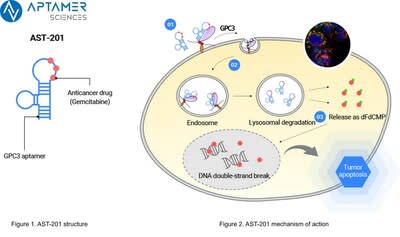
Fig 1. AST-201 structure (Among the sequence of GPC3 aptamer, the payload of gemcitabine is conjugated as the part of sequence.)
Fig 2. AST-201 mechanism of action
1. AST-201 targets and binds to the GPC3 protein located on the surface of cancer cells.
2. The drug undergoes internalization into the cancer cell by endocytosis.
3. After the lysosomal degradation, gemcitabine monophosphate (dFdCMP), the active form of gemcitabine, is released within the cell leading to programmed cell death of the tumor cell (apoptosis).
About Aptamer Sciences, Inc.
Aptamer Sciences Inc., listed on KOSDAQ in South Korea, is a global leading company in aptamer field. The company has established an integrated aptamer technology platform, called ApDCTM (Aptamer Drug Conjugate) and focused on developing oncology pipelines.
For more information, please visit the website, www.aptsci.com/en or contact bd@aptsci.com.


View original content to download multimedia:https://www.prnewswire.com/news-releases/aptamer-sciences-inc-files-ind-application-for-innovative-liver-cancer-treatment-ast-201-a-promising-approach-to-address-under-medical-needs-302098802.html
SOURCE Aptamer Sciences Inc.