Bausch Health (BHC) Ulcerative Colitis Drug Meets Primary Goal
Bausch Health Companies Inc. BHC announced positive top-line results from the mid-stage study evaluating amiselimod for the treatment of ulcerative colitis (“UC”).
Amiselimod is a sphingosine-1-phosphate (“S1P”) receptor functional antagonist and, by inhibiting the receptor function of the lymphocyte S1P receptor, retains lymphocytes sequestered in the lymph nodes and prevents them from contributing to autoimmune reactions.
The phase II study was a 12-week, double-blind, placebo-controlled, randomized, dose-ranging study to evaluate the efficacy and safety of amiselimod in 320 patients with mildly-to-moderately active UC.
Amiselimod met the primary and key secondary endpoints, including clinical and endoscopic measures, in the double-blind period of the study. The primary endpoint, change in modified Mayo Score at Day 85, was -2.3 for amiselimod compared with placebo (-1.6). Data showed that 32.4% of patients on amiselimod achieved clinical remission compared with 17.8% on placebo, while 42.7% of patients on amiselimod achieved endoscopic improvement versus only 23.4% on placebo.
The open-label extension of up to 52 weeks is currently ongoing. Efficacy results were similar for both dose groups (0.2 mg and 0.4 mg). The candidate was well-tolerated, with no unexpected adverse events.
The results of the previous study evaluating the cardiac effects of amiselimod, along with this phase II study results, show that the candidate has a safety profile.
The successful development of the candidate will diversify Bausch’s gastrointestinal business, Salix.
The stock has gained 18.8% year to date compared with the industry’s growth of 36.6%.
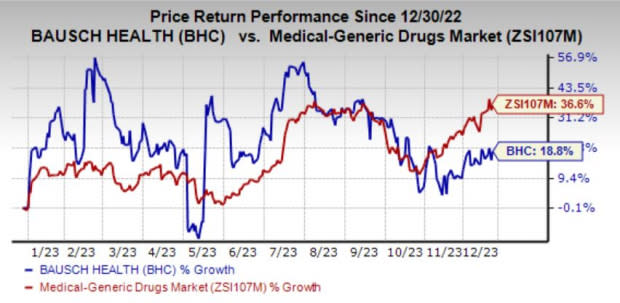
Image Source: Zacks Investment Research
Salix maintains momentum as Xifaxan, Relistor and Trulance continue to grow. Robust sales of Xifaxan fueled the top line and the drug is one of the top growth drivers of the company.
Meanwhile, two global phase III studies for the use of a rifaximin soluble solid dispersion formulation for the prevention of overt hepatic encephalopathy in patients with early decompensation in liver cirrhosis have commenced. Bausch Health expects to complete enrollment of two global phase III studies in the first quarter of 2024.
However, the neurology and dermatology businesses continue to face long-term challenges that will impact the Diversified segment.
Bausch is currently in a transitional mode with the separation of its eye health business into an independent public company. The move should improve strategic focus and enhance financial transparency. Its efforts to reduce its massive debt are also encouraging.
Zacks Rank and Other Stocks to Consider
Bausch currently carries a Zacks Rank #2 (Buy).
A few other top-ranked stocks in the overall sector are CytomX Therapeutics, Inc. CTMX, Entrada Therapeutics, Inc. TRDA and Puma Biotechnology, Inc. PBYI each sporting a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
In the past 60 days, estimates for CytomX Therapeutics’ 2023 bottom line have improved from a loss of 37 cents per share to earnings of 2 cents. Meanwhile, loss per share estimates for 2024 have narrowed from 51 cents to 6 cents. Year to date, shares of CTMX have lost 11.8%.
Estimates for Entrada’s 2023 loss per share have narrowed from $2.07 to 9 cents in the past 60 days. The same for 2024 has narrowed from $2.35 to $2.04 during the same time frame. Year to date, shares of TRDA have gained 9.2%. Earnings of Entrada Therapeutics beat estimates in three of the last four quarters while missing the same on the remaining occasion.
In the past 60 days, estimates for Puma Biotechnology’s 2023 earnings per share have improved from 67 cents to 72 cents. During the same period, earnings per share estimates for 2024 have moved up from 56 cents to 62 cents. Year to date, shares of PBYI have lost 6.9%.
Earnings of Puma Biotechnology beat estimates in three of the last four quarters while missing the same on the remaining occasion. PBYI delivered a four-quarter average earnings surprise of 76.55%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Puma Biotechnology, Inc. (PBYI) : Free Stock Analysis Report
CytomX Therapeutics, Inc. (CTMX) : Free Stock Analysis Report
Bausch Health Cos Inc. (BHC) : Free Stock Analysis Report
Entrada Therapeutics, Inc. (TRDA) : Free Stock Analysis Report
