Biotech Stock On The Radar: Anavex, A Neurology-Focused Precision Medicine Company

The European Rett Syndrome Conference gets underway Friday in Finland. Among the presenters: a New York-based micro-cap biotech that has an investigational asset to treat Rett syndrome in its pipeline.
Anavex Life Sciences Corp (NASDAQ: AVXL), founded in 2006, is a biotech that focuses on developing therapies for neurodegenerative and neurodevelopmental disorders.
Anavex's proprietary SIGMARECEPTOR Discovery platform is used to produce small molecule drug candidates with unique modes of action.
The lead compound ANAVEX 2-73, generically blarcamesine, is being developed for Alzheimer's, Parkinson's disease and other central nervous system diseases, including rare diseases such as Rett syndrome.
Apart from ANAVEX 2-73, it has four pipeline assets in various stages of development that are being evaluated for multiple indications such as Alzheimer's, Parkinson's, pain and cancer.
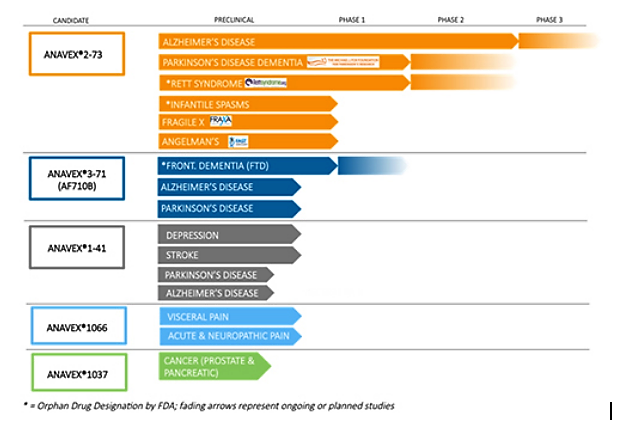
Source: Anavex
ANAVEX 2-73: Mechanism, Progress, Prospects
Neurological diseases are often caused by chronic homeostatic imbalances or cellular stress of brain cells. This could be due to the constant genetic mutation in pediatric diseases such as Rett syndrome and infantile spasms, and by the age-correlated buildup of cellular insult in neurodegenerative diseases such as Alzheimer's and Parkinson's.
When the SIGMAR1 gene — which encodes the S1R protein responsible for the restoring of homeostasis and the recovery of cell function — is activated by SIGMAR1 agonists like ANAVEX 2-73, it helps restore cellular balance, or homeostasis.
The following are the ongoing trials for ANAVEX 2-73:
Enrollment of the Phase 2b/3 study in Alzheimer's is ongoing; a voluntary extension study is also planned.
Enrollments for the U.S. Phase 2 study in Rett syndrome and the AVATAR Rett study are proceeding. Recently, the Independent Data Safety Monitoring Board which reviewed the U.S. Phase 2 safety and efficacy data recommended continuation of the study without modification.
Enrollment of Phase 2 study in PD is nearing completion; a voluntary extension study is also planned.
For the other indications — Fragile X syndrome, Angelman syndrome and infantile spasms — ANAVEX 2-73 is being evaluated in preclinical trials.
On Sept. 16, Anavex released preliminary data from the first six-patient cohort in the Phase 2 Rett syndrome trial, which evaluated ANAVEX 2-73 in patients ages 18-36 who received a low dose of about 5 mg daily oral liquid dose for seven weeks.
Rett syndrome is a rare brain condition caused by mutations in the MECP2 gene. Since the gene is found on the X chromosome, Rett syndrome occurs mostly in girls. It affects about one in 10,000-15,000 females.
The company said both efficacy endpoints, the Rett Syndrome Behavior Questionnaire and the Clinical Global Impression Improvement, showed significant baseline improvement.
Additionally, the plasma levels of the biomarker glutamate also declined significantly.
Biomarkers are increasingly used in CNS studies to confirm target and pathway engagement.
Anavex is due to present detailed Phase 2 data at the Rett syndrome conference Friday.
"We believe the data presentation could include the PK data and data from the placebo-treated patients, Janney analyst Yun Zhong said in a recent note.
"We continue to be optimistic that ANAVEX 2-73, by activating the Sigma-1 receptor that is involved in multiple pathways, could demonstrate efficacy in neurodegenerative and neurodevelopmental disorders."
See Also: Axsome Shares Flirt With Record High: How Much Upside Is Left?
Competition, Market Potential
Anavex's competitors include big pharma companies such as Biogen Inc (NASDAQ: BIIB), Pfizer Inc. (NYSE: PFE), Novartis AG (NYSE: NVS), Merck & Co., Inc. (NYSE: MRK), Eli Lilly And Co (NYSE: LLY), Johnson & Johnson (NYSE: JNJ), Roche Holdings AG Basel ADR (OTC: RHHBY), GlaxoSmithKline plc (NYSE: GSK) and Allergan plc (NYSE: AGN), as well as Axovant Gene Therapies Ltd (NASDAQ: AXGT) and Perrigo Company PLC (NYSE: PRGO).
Alzheimer's is likely to afflict about 7.1 million Americans by 2025, representing a 25% increase from today's incidence rate.
As existing medications treat only symptoms and are not effective in stopping the onset or progression of the disease, there is an acute need for an effective drug.
Alzheimer's drug development is considered to have the highest failure rate of any drug research.
Parkinson's disease also brings a multimillion-dollar opportunity. The PD market is set to expand from $2.1 billion in 2014 to $3.2 billion by 2021, Avanex said, citing estimates from GBI Research.
The Finances
Anavex has yet to post any revenue. For the fiscal year ended Sept. 30, 2018, the company reported a loss of 39 cents per share versus a loss of 33 cents in 2017.
For the third quarter ended June 30, 2019, the company reported a loss of 14 cents per share, more than double the year-ago loss of 6 cents per share. R&D expenses ballooned about 92% to $7.15 million.
Roughly 81% of the operating expenses were for R&D, and the remainder went toward meeting general and administrative expenses.
At the end of the June quarter of 2019, Anavex had cash and cash equivalents of $21.25 million, slightly lower than the $22.93 million it held at the end of the September quarter of 2018.
The cash and cash equivalents as well as financial commitments from third parties, are sufficient to meet cash needs for over two years, according to the company.
Stock Take
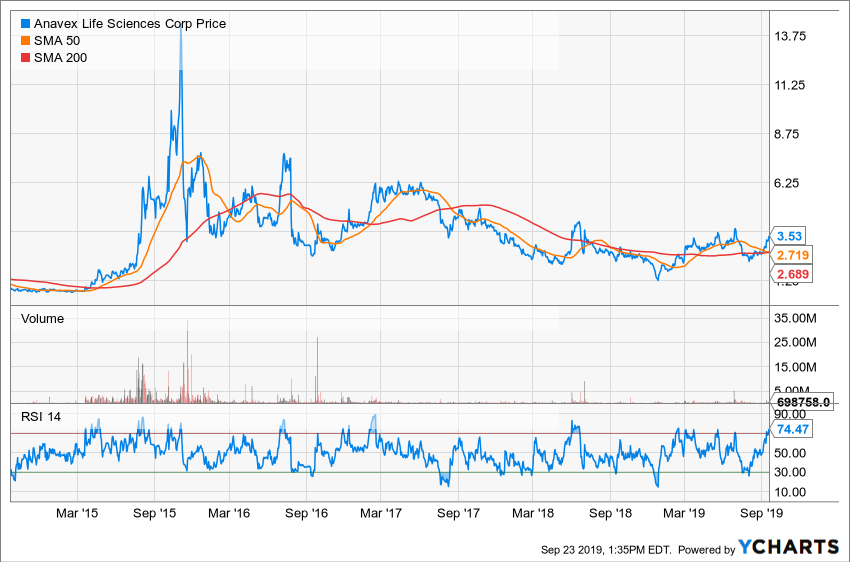
Anavex shares have risen about 126% year-to-date. The $3.75-$4.25 region has intermittently served as a resistance and support level in the past. With the stock trading around $3.53, a climb to the $3.75-$4.25 area could trigger some selling.
If shares find enough buyers to breach this level, the $6.50-$7.50 area could serve as next resistance zone. On the downside, the $2.40 level is likely to offer support to the stock.
Source: Y Charts
Roughly 13% of the float is being held as short, with the short ratio at 14.4.
Although not alarming, short interest has been steadily climbing. This portends the risk of a short squeeze, which could send the shares sharply higher in the wake of positive news.
Janney analyst Zhong has a Buy rating on Avanex Life Sciences with a $10 price target for the shares, suggesting roughly 180% upside potential.
Latest Ratings for AVXL
Jul 2019 | Initiates Coverage On | Buy | ||
Jun 2019 | Initiates Coverage On | Buy | ||
May 2018 | Initiates Coverage On | Buy |
View More Analyst Ratings for AVXL
View the Latest Analyst Ratings
See more from Benzinga
© 2019 Benzinga.com. Benzinga does not provide investment advice. All rights reserved.