Glaxo (GSK)/Vir Affirm COVID-19 Drug Cuts Hospitalization/Death
GlaxoSmithKline GSK and partner Vir Biotech VIR announced final day 29 data from the COMET- ICE study, which confirmed that their monoclonal antibody treatment, sotrovimab, significantly reduces hospitalization and risk of death in adults with mild-to-moderate COVID-19 who are at high risk of progression to severe disease. Data from a primary efficacy analysis of all 1,057 patients in the study demonstrated a 79% reduction in hospitalization for more than 24 hours or death compared with placebo after 29 days.
Sotrovimab was granted emergency approval by the FDA last month for treating mild-to-moderate COVID-19 in patients who are at risk of progression to severe COVID-19, including hospitalization or death. The EUA was based on an interim analysis of efficacy and safety data from the COMET-ICE study, which demonstrated an 85% reduction in hospitalization or death on treatment VIR-7831. The latest safety and efficacy data from the study at day 29 for the full population validates the interim efficacy data from the study.
Additionally, the company announced that the U.S. National Institutes of Health (NIH) updated its COVID-19 treatment guidelines to recommend use of sotrovimab. The companies also said they have approximately 450,000 doses of sotrovimab in hand for supply in the United States.
The companies plan to submit a formal biologics license application (BLA), seeking approval of sotrovimab in the second half of 2021.
So far this year, Glaxo’s shares have risen 8.1% compared with the industry’s 7% increase.
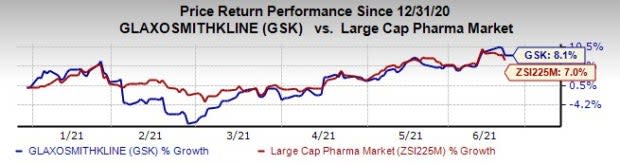
Image Source: Zacks Investment Research
Meanwhile the European Medicines Agency (EMA) has also started a rolling review of data on sotrovimab. The rolling review will continue until enough data is obtained to support a formal filing of marketing authorization application in the EU. In May, the European Medicines Agency’s (EMA) Committee for Human Medicinal Products (CHMP) also issued a positive scientific opinion on sotrovimab for the early treatment of COVID-19.
In a separate press release, ViiV Healthcare announced that it has signed a global collaboration with Halozyme Therapeutics HALO to leverage the latter’s ENHANZE drug delivery technology to enable development of “ultra long-acting” medicines for HIV treatment and prevention. Glaxo will utilize Halozyme’s technology to administer large volume subcutaneous injections of its long-acting injectables, which in turn may further extend dosing intervals for ViiV’s pipeline of HIV medicines.
Per the deal, ViiV Healthcare has acquired an exclusive license to four HIV small and large molecule targets for which it will make an upfront payment of $40 million. Halozyme will also be entitled to development and commercial milestone payments of up to $175 million per target.
ViiV Healthcare plans to begin the first trial with the technology by the end of 2021 on its candidate, long-acting cabotegravir for prevention of HIV, also called pre-exposure prophylaxis, or PrEP. We remind investors that ViiV Healthcare has begun rolling submission of new drug application with the FDA for its long-acting cabotegravir for PrEP.
ViiV Healthcare is majorly owned by Glaxo with Pfizer PFE and Shionogi Limited having stake in the company.
Glaxo currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
5 Stocks Set to Double
Each was hand-picked by a Zacks expert as the #1 favorite stock to gain +100% or more in 2020. Each comes from a different sector and has unique qualities and catalysts that could fuel exceptional growth.
Most of the stocks in this report are flying under Wall Street radar, which provides a great opportunity to get in on the ground floor.
Today, See These 5 Potential Home Runs >>
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
GlaxoSmithKline plc (GSK) : Free Stock Analysis Report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Halozyme Therapeutics, Inc. (HALO) : Free Stock Analysis Report
Vir Biotechnology, Inc. (VIR) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research
