Juul surpasses Facebook as fastest startup to reach decacorn status
If it seems like seeing people vape on devices that look like USB flash drives came seemingly out of nowhere, you’re absolutely right.
It’s the ubiquity of the e-cigarette, known as the Juul, that has led its manufacturer to achieve a milestone faster than any other startup in history – including Facebook.

With its latest fundraising round first reported by Bloomberg this summer, Juul Labs crossed the decacorn threshold (a valuation of over $10 billion) by earning a $15 billion valuation just seven months after its first VC round. According to funding data compiled by Pitchbook for Yahoo Finance, that’s four-times faster than Facebook (FB), nearly five-times faster than Snapchat (SNAP) and 11-times faster than Dropbox (DBX).
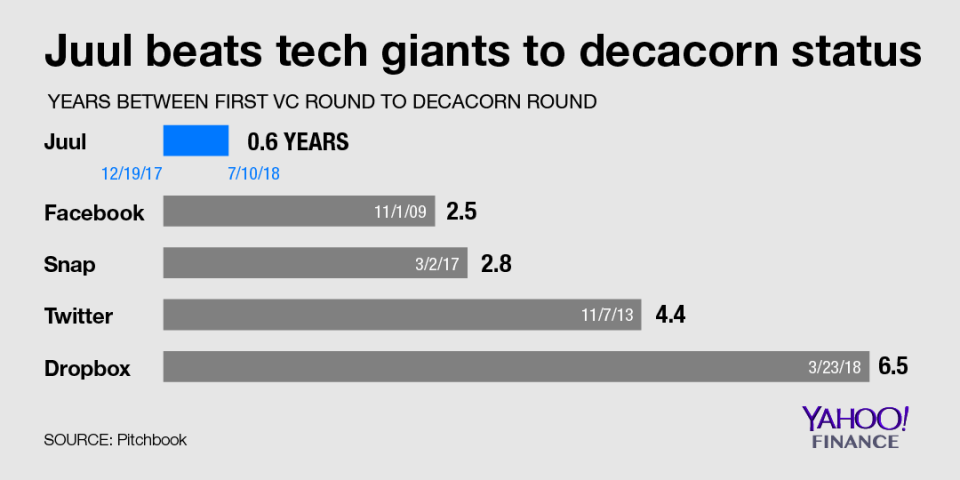
It’s an incredible feat for a company that, unlike most of the other decacorns, actually sells a physical product and as of just three years ago didn’t even exist. Pax Labs, which originally launched the Juul with a team of 20 and just $2 million, spun off Juul Labs in 2017 after the e-cigarette’s debut in 2015. The San Francisco-based startup then raised $111 million in its first round as a separate entity in December 2017.
Since then, Juul has been on a tear to dominate the e-cigarette market. Its latest fundraising round in July, led by a $650 million investment from Tiger Global Management, came on the heels of capturing over 60% of total e-cigarette sales, according to Nielsen data cited by Wells Fargo analyst Bonnie Herzog. As of September, Herzog’s same report showed Juul’s 52-week sales grew 770% over the same period a year ago to top $1.47 billion, representing nearly 73% of the entire category.
That incredible clip helps explain how Juul was able to reach decacorn status just seven months after its first VC round, compared to the five-year average posted by its decacorn peers. Juul’s decacorn round deal size also notably dwarfs others in the club. The $1.25 billion the company said it planned to raise in a 2018 Securities and Exchange Commission filing is about four-times as much as the amount raised in Facebook’s, Snap’s (SNAP) and Twitter’s (TWTR) respective decacorn rounds.

A decacorn targeted by the FDA
Juul’s growth, however, has been mired by increasing scrutiny over how it may attract underage customers, and with the company’s meteoric rise has come increased attention from the Food & Drug Administration. Just last week, the FDA announced it conducted an “unannounced on-site inspection” at Juul Labs headquarters in San Francisco.
Earlier in September, the FDA ramped up efforts to curtail underage use of e-cigarettes by requiring Juul and other e-cigarette makers to share a plan on how they will prevent teenagers from using their products. Juul has 60 days to comply with the FDA request.
In Juul’s hometown, San Francisco voters have also been adding regulatory pressure on the firm. The city approved a ban on flavored nicotine products, including e-cigarettes, in June. While flavored cigarettes have been banned by the FDA for years due to their well-studied appeal to younger users, the agency has yet to extend the same ban unilaterally to e-cigarettes. Earlier this year, Juul changed some of its flavor names, including replacing “cool cucumber” and “creme brulee” with “cucumber” and “creme.”
Whether Juul’s breakneck growth will be impeded by looming FDA regulations remains to be seen. For now, though, the startup wears the crown as the fastest decacorn of all.
Zack Guzman is a reporter for Yahoo Finance. Follow him on Twitter @zGuz.
Read more:
