Prothena (PRTA) Q2 Earnings and Revenues Miss Estimates
Prothena Corporation PRTA reported a second-quarter 2023 loss of $1.03 per share, wider than the Zacks Consensus Estimate of a loss of 92 cents per share. In the year-ago quarter, the clinical-stage company had reported a loss of 88 cents per share.
Higher expenses impacted the bottom line.
Total revenues were $4 million in the second quarter, which lagged the Zacks Consensus Estimate of $11 million. In the year-ago quarter, the company had recorded total revenues of $1.3 million. Revenues in the reported quarter primarily included collaboration revenues from Bristol-Myers Squibb BMY.
The company’s shares have rallied 10.5% year to date against the industry’s decline of 13.1%.
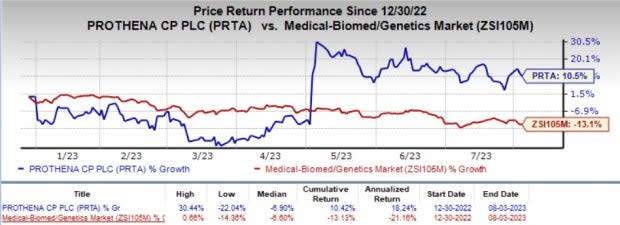
Image Source: Zacks Investment Research
Quarter in Detail
Collaboration revenues beat our model estimate of $3 million.
Research and development (R&D) expenses totaled $56 million, up 77.2% year over year primarily due to higher clinical study expenses, rising personnel-related expenses, and increased consulting and other R&D expenses.
General and administrative (G&A) expenses amounted to $14.5 million, up from $13 million in the year-ago quarter. This was primarily due to higher personnel-related and consulting expenses.
As of Jun 30, 2022, Prothena had $661.3 million in cash, cash equivalents and restricted cash compared with $688.4 million as of Mar 31, 2023. It had no debt as of the same date.
Updated 2023 Guidance
The company expects its 2023 net cash burn from operating and investing activities in the range of $148-$161 million (compared with the previous $213-$229 million range).
Prothena expects the year-end cash, cash equivalents and restricted cash midpoint to be approximately $600 million (compared with the previously projected $512 million). The rise reflects $55 million exclusive worldwide rights deal for PRX005 made with BMY.
Net loss for 2023 is expected in the band of $153-$171 million (compared with the previous $250-$275 million range).
Pipeline Updates
Prothena is advancing an early-stage pipeline of programs for a number of potential neurological indications with Bristol-Myers. PRX005 — a potential treatment for Alzheimer’s Disease (AD) — is an investigational antibody that targets tau, a protein implicated in diseases including AD, frontotemporal dementia, progressive supranuclear palsy, chronic traumatic encephalopathy and other tauopathies.
The company reported the top-line data from the phase I single ascending dose (SAD) study in January 2023, which showed that single doses of PRX005 across three dose cohorts were generally safe and well tolerated, meeting the primary objective of the study. The top-line data from the ongoing phase I multi ascending dose (MAD) study is yet to be reported by BMY.
In July, BMY exercised an option to obtain exclusive worldwide commercial rights for PRX005. Under the terms of the deal, BMY will pay $55 million to Prothena for the license. BMY is responsible for future development, manufacturing and commercialization of the candidate.
Prothena is evaluating PRX012 — an investigational high-potency monoclonal antibody targeting a key epitope at the N-terminus of amyloid beta (Aβ) — for treating AD. Phase I SAD and MAD studies in healthy volunteers and patients with AD are ongoing. The top-line data from these studies is expected later in 2023.
The company is also developing a dual Aβ-Tau vaccine — a potential prevention and treatment for AD — to target key epitopes within Aβ and tau proteins to promote amyloid clearance and block pathogenic tau interaction. An investigational new drug application filing for the vaccine is anticipated by the end of 2023.
The company is evaluating prasinezumab in collaboration with Roche RHHBY for the treatment of Parkinson’s disease (PD). In the first quarter, Roche completed the enrollment for the phase IIb PADOVA study in patients with early PD (NCT04777331) and the top-line data is expected in 2024.
Prothena is also evaluating birtamimab, a potential treatment for AL amyloidosis. In 2021, it reached a Special Protocol Assessment agreement with the FDA and initiated a confirmatory phase III AFFIRM-AL study of birtamimab in Mayo Stage IV patients with AL amyloidosis. Result from the study is expected in 2024.
Novo Nordisk NVO acquired Prothena’s clinical-stage antibody, PRX004 (now NNC6019) and a broader ATTR amyloidosis program. The company received a $40-million milestone payment from Novo Nordisk in December 2022 related to the continued advancement of NNC6019 in a phase II study.
Novo Nordisk is conducting a phase II study in patients with ATTR cardiomyopathy and the top-line data from the same is expected in 2024.
Prothena Corporation plc Price and Consensus
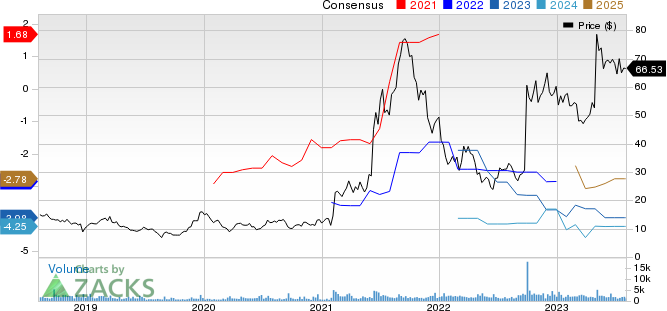
Prothena Corporation plc price-consensus-chart | Prothena Corporation plc Quote
Zacks Rank
Prothena currently carries a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Roche Holding AG (RHHBY) : Free Stock Analysis Report
Bristol Myers Squibb Company (BMY) : Free Stock Analysis Report
Novo Nordisk A/S (NVO) : Free Stock Analysis Report
Prothena Corporation plc (PRTA) : Free Stock Analysis Report
