NasdaqGS - Nasdaq Real Time Price • USD
Gilead Sciences, Inc. (GILD)
At close: April 26 at 4:00 PM EDT
After hours: April 26 at 7:55 PM EDT
All SEC Filings
Corporate Changes & Voting Matters
Periodic Financial Reports
Proxy Statements
Tender Offer/Acquisition Reports
Offering Registrations
-

-

-
Q1 2024 Gilead Sciences Inc Earnings Call
Q1 2024 Gilead Sciences Inc Earnings Call
Thomson Reuters StreetEvents • 12 hours ago -

-
-

-

-

-
-

-

-

-

-

-
-

-

-
-

-

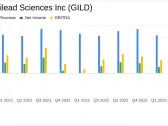
Performance Overview
Trailing total returns as of 4/26/2024, which may include dividends or other distributions. Benchmark is S&P 500
| Return | GILD | S&P 500 |
|---|---|---|
| YTD | -18.42% | +6.92% |
| 1-Year | -20.56% | +25.26% |
| 3-Year | +13.01% | +22.00% |